1. Bond Length:
- It is the equilibrium distance between the nuclei of two bonded atoms in a molecule.
- The covalent radius is the radius of an atom’s core which is in contact with the core of an adjacent atom in a bonded situation.
- If two similar atoms are joined by a covalent bond in the same molecule, the covalent radius is half of the distance between them.
- The van der Waals radius represents the overall size of the atom which includes its valence shell in a non-bonded situation. The van der Waals radius is half of the distance between two similar atoms in separate molecules in a solid.
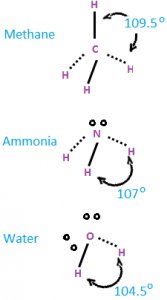
2. Bond Angle:
- It is the angle between the orbitals containing bonding electron pairs around the central atom in a molecule/complex ion.
3. Bond Enthalpy:
- It is defined as the amount of energy required to break one mole of bonds of a particular type between two atoms in a gaseous state.
- The unit of bond enthalpy is kJ mol–1.
- Larger the bond dissociation enthalpy, stronger will be the bond in the molecule.
- In polyatomic molecules the bond enthalpy value for breaking all the bonds is not same. So, the term mean or average bond enthalpy is used. It is obtained by dividing total bond dissociation enthalpy by the number of bonds broken.
Ex: H2O (g) → H (g) + OH (g); ΔH–1 = 502 kJ / mol
OH (g) → H (g) + O (g); ΔH–2 = 427 kJ/mol
Average bond enthalpy = 502 + 427/2 = 464.5 kJ/mol
4. Bond Order:
- In the Lewis description of covalent bond, the Bond Order is given by the number of bonds between the two atoms in a molecule.
Ex: Bond order in Cl2 = 1
Bond order in O2 = 2 - Isoelectronic molecules and ions have identical bond orders; for example, F2 and O2-2 have bond order 1. N2, CO and NO+ have bond order 3.
- With increase in bond order, bond enthalpy increases and bond length decreases.
5. Resonance Structures:
- A single Lewis structure is inadequate for the representation of a molecule in conformity with its experimentally determined parameters.
- According to the concept of resonance, whenever a single Lewis structure cannot describe a molecule accurately, a number of structures with similar energy, positions of nuclei, bonding and non-bonding pairs of electrons are taken as the canonical structures of the hybrid which describes the molecule accurately.
- This is also called resonance hybrid. Resonance is represented by a double headed arrow.
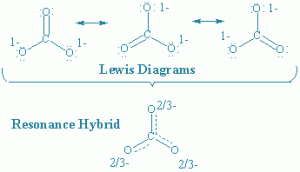
- Resonance stabilizes the molecule as the energy of the resonance hybrid is less than the energy of any single canonical structure.
- Resonance averages the bond characteristics as a whole
