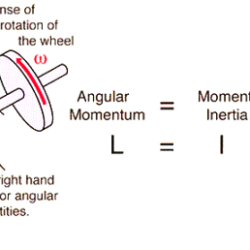
Relation between Angular Momentum and Moment of inertia
When a body moves along a straight line, then the product of the, mass m and the linear velocity of the body is called the “linear momentum” p of the body (p = m*v), if a body is rotating about an axis, then the sum of the moments of the linear momenta of all the Read more about Relation between Angular Momentum and Moment of inertia[…]