Statement: “Energy can neither be created nor be destroyed. But it can be converted from one form of energy into another form of energy.”
When a system changes for a given initial state to a given final state, both the work W and Q depend on the nature of the process.
Experimentally, however, we find a surprising thing, the quantities Q and W is the same for all processes. It depends only on initial and final states and does not depend at all on how the system gets one state form the. All other combination of Q and W including Q alone, W alone, Q + W and Q – 2W are path dependent, only the quantity Q – W is not.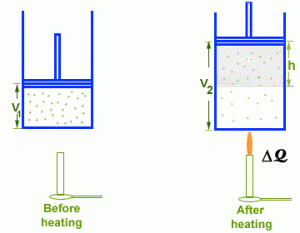 Where, V₁ = Initial volume
Where, V₁ = Initial volume
V₂ = Final Volume
The volume of system increases from V₁ to V₂
The quantity Q – W must represent a change in same intrinsic property of the system. This property is the internal energy U and we write
ΔU = Uf – Uᵢ = Q – W
This equation is the first law of thermodynamics.
If the thermodynamic system undergoes only a differential change, we can write
dU = dQ – dW
We can write this equation as dQ = dU + dW
Thus, we can say that, heat supplied to the system is the sum of external work done by the system and increase in its internal energy.
