⇒ The oxoacids of sulphur are more numerous and more important than those of Se and Te.
⇒ Many of the oxoacids of sulphur do not exist as free acids, but are known as anions and salts.
⇒ Acids ending in ous have S in the oxidation state (+IV) and form salts in -ite.
⇒ Acids ending in -ic have S in the oxidation state (+VI) and form salts ending in -ate.
|
Acids |
Structure | Oxidation state of S |
Important properties |
|
Sulphurous acids |
|||
|
H₂SO₃ – sulophurous acid |
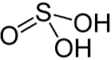 |
+4 |
SO₂ + H₂O → H₂SO₃ |
|
H₂S₂O₅ – Di/pyrosulphurous acid |
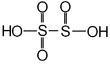 |
Sulfur atom bonded to 3 oxygen atoms: +5 The other sulfur atom: +3 |
Does not exist in the free state |
|
H₂S₂O₄ – Dithionous acid |
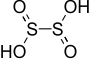 |
+3 |
|
|
Sulphuric acid series |
|||
|
H₂SO₄ – Sulphuric acid |
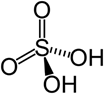 |
+6 |
Highly corrosive strong mineral acid with the molecular formula H₂SO₄. |
|
H₂S₂O₃ – Thiosulphuric acid |
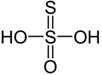 |
+2 |
Aqueous solutions decompose. |
|
H₂S₂O₇ – Disulfuric acid (Pyrosulphuric acid) |
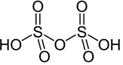 |
+6 |
Strong oxidizing agent |
|
H₂SO₅ – Peroxymono sulphuric acid |
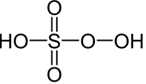 |
+6 |
H₂O₂ + H₂SO4 ⇌ H₂SO₅ + H₂O |
| H₂S₂O₈ – Peroxydisulfuric acid | 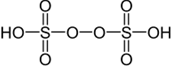 |
+6 |
2ClSO3H + H₂O₂ → H₂S₂O₈ + 2HCl |
|
Thionic acids |
|||
| H₂S₂O₆ – Dithionic acid | 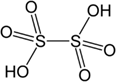 |
+5
|
Not isolated in pure form, only concentrated solutions have been prepared |
|
H₂SnO₆ – Polythionic acid (n = 1 – 12) |
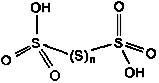 |
0 (for the bridging S atoms), +5 (for the terminal central S atoms) |
|
Sodium thiosulfate:
⇒ Sodium thiosulfate (Na₂S₂O₃) is an inorganic compound that is typically available as the pentahydrate, Na₂S₂O₃·5H₂O.
⇒ The solid is an efflorescent (loses water readily) crystalline substance that dissolves well in water.
⇒ It is also called sodium hyposulfite or “hypo”
Industrial production and laboratory synthesis:
In the laboratory, this salt can be prepared by heating an aqueous solution of sodium sulfite with sulfur or by boiling aqueous sodium hydroxide and sulfur according to this equation:
6 NaOH + 4 S → 2 Na₂S + Na₂S₂O₃ + 3 H₂O
Principal reactions:
⇒ Upon heating to 300 °C, it decomposes to sodium sulfate and sodium polysulfide:
4 Na₂S₂O₃ → 3 Na₂SO4 + Na₂S5
⇒ Under normal conditions, acidification of solutions of this salt excess with even dilute acids results in complete decomposition to sulfur, sulfur dioxide, and water:
Na₂S₂O₃ + 2 HCl → 2 NaCl + S + SO₂ + H₂O
Uses:
⇒ Iodometry: The thiosulfate anion reacts stoichiometrically with iodine in aqueous solution, reducing it to iodide as it is oxidized to tetrathionate:
2 S₂O₃²⁻ + I₂ → S₄O₆²⁻ + 2 I⁻
⇒ Photographic processing: Silver halides, e.g., AgBr, typical components of photographic emulsions, dissolve upon treatment with aqueous thiosulfate:
2 S₂O₃²⁻ + AgBr → [Ag (S₂O₃)₂]³⁻ + Br⁻
⇒ Aluminium cation reaction: When heated with a sample containing aluminium cations it produces a white precipitate:
2 Al³⁺ + 3 S₂O₃²⁻ + 3 H₂O → 3 SO₂ + 3 S + 2 Al (OH)3
⇒ Organic chemistry: Alkylation of sodium thiosulphate gives S-alkylthiosulfonates, which are called Bunte salts. This reaction is employed in one synthesis of the industrial reagent thioglycolic acid:
ClCH₂CO₂H + Na₂S₂O₃ → Na [O₃S₂CH₂CO₂H] + NaCl
Na [O₃S₂CH₂CO₂H] + H₂O → HSCH₂CO₂H + NaHSO₄
⇒ Neutralizing bleach, chlorinated water, and related treatments: It is used to dechlorinate tap water including lowering chlorine levels for use in aquaria and swimming pools and spas and within water treatment plants to treat settled backwash water prior to release into rivers. The reduction reaction is analogous to the iodine reduction reaction.
Thiosulfate reduces the hypochlorite (active ingredient in bleach) and in so doing becomes oxidized to sulfate. The complete reaction is:
4 NaClO + Na₂S₂O₃ + 2 NaOH → 4 NaCl + 2 Na₂SO₄ + H₂O
