- Introduced by Pauling
- The atomic orbitals combine to form new set of equivalent orbitals known as hybrid orbitals.
- Unlike pure orbitals, the hybrid orbitals are used in bond formation.
- Hybridization: The process of intermixing of the orbitals of slightly different energies so as to redistribute their energies, resulting in the formation of new set of orbitals of equivalent energies and shape.
Features of hybridization:
- The number of hybrid orbitals is equal to the number of the atomic orbitals that get hybridised.
- The hybridised orbitals are always equivalent in energy and shape.
- The hybrid orbitals are more effective in forming stable bonds than the pure atomic orbitals.
- These hybrid orbitals are directed in space in some preferred direction to have minimum repulsion between electron pairs and thus a stable arrangement. Therefore, the type of hybridization indicates the geometry of the molecules.
Important conditions for hybridisation:
- The orbitals present in the valence shell of the atom are hybridised
- The orbitals undergoing hybridization should have almost equal energy.
- Promotion of electron is not essential condition prior to hybridisation.
- It is not necessary that only half filled orbitals participate in hybridisation. In some cases, even filled orbitals of valence shell take part in hybridisation.
Types of hybridization:
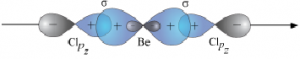
sp hybridisation:
- One s and one p orbitals are mixed to form two equivalent sp hybrid orbitals.
- Each sp hybrid orbitals has 50% s-character and 50% p-character.
- This type of hybridisation is also known as diagonal hybridisation.
- Such a molecule possesses linear geometry.
- The two sp hybrids point in the opposite direction along an axis with projecting positive lobes and very small negative lobes, which provides more effective overlapping resulting in the formation of stronger bonds.
Ex: BeCl₂
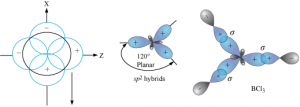
sp² hybridisation:
- One s and two p-orbitals are mixed to form three equivalent sp² hybridised orbitals.
- The three hybrid orbitals so formed are oriented in a trigonal planar arrangement.
Ex: BCl₃
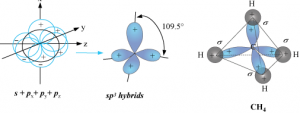
sp³ hybridisation:
- One s-orbital and three p-orbitals are mixed to form four sp³ hybrid orbital of equivalent energies and shape.
- There is 25% s-character and 75% p-character in each sp³ hybrid orbital.
- The four sp³ hybrid orbitals so formed are directed towards the four corners of the tetrahedron. The angle between sp³ hybrid orbital is 109.5°.
Ex: CH₄
Ex: NH₃ Molecule:
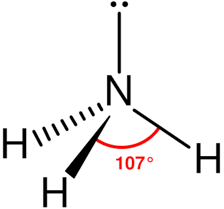
Note: Here, N has one lone pair so, its shape is pyramidal.
