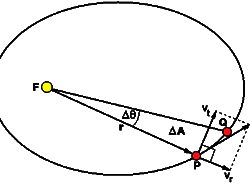
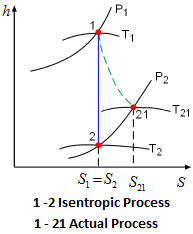
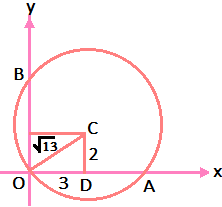
Division Algorithm
Division Algorithm If 0 ≠ a, b ϵ Z then ∃ q, r ϵ Z uniquely such that b = aq + r where 0 ≤ r < |a|. A nonzero integer a is said to divide an integer b if there exist q ϵ z such that b = aq. It is denoted by Read more about Division Algorithm[…]