Hybridisation of Elements involving d Orbitals
- The elements which contain d orbitals in addition to s and p orbitals form hybrid orbitals with s, p and d orbitals.
- The hybridisation involves either [3s, 3p and 3d] or [3d, 4s and 4p] (as the energies of these orbitals are comparable).
- However, since the difference in energies of 3p and 4s orbitals is significant, no hybridisation involving 3p, 3d and 4s orbitals is possible.
|
Type of hybridisation |
Atomic Orbitals Involved |
Spatial Orientation of Hybrid Orbitals |
|
sp³d |
s + \(\underbrace{~{{p}_{x}}+\text{ }{{p}_{y}}+\text{ }{{p}_{z}}}_{{}}\)+ \(\underbrace{{{d}_{z}}_{{}^\text{2}}}_{outer}\) |
Trigonal Bipyramidal |
|
d sp³ |
\(\underbrace{{{d}_{z}}_{{}^\text{2}}}_{Inner}\)+ s + \(\underbrace{~{{p}_{x}}+\text{ }{{p}_{y}}+\text{ }{{p}_{z}}}_{{}}\) |
Trigonal Bipyramidal |
|
sp³ d² |
s + \(\underbrace{~{{p}_{x}}+\text{ }{{p}_{y}}+\text{ }{{p}_{z}}}_{{}}\) +\(\underbrace{{{d}_{z}}_{{}^\text{2}}+{{d}_{x}}_{{}^\text{2}-\text{ }y{}^\text{2}}}_{outer}\) |
Octahedral |
|
d²sp³ |
\(\underbrace{{{d}_{z}}_{{}^\text{2}}+{{d}_{x}}_{{}^\text{2}-\text{ }y{}^\text{2}}}_{Inner}\)+ s + \(\underbrace{~{{p}_{x}}+\text{ }{{p}_{y}}+\text{ }{{p}_{z}}}_{{}}\) |
Octahedral |
|
dsp² |
\(\underbrace{{{d}_{x}}_{{}^\text{2}-\text{ }y{}^\text{2}}}_{Inner}\)+ s +\(\underbrace{~{{p}_{x}}+\text{ }{{p}_{y}}}_{{}}\) |
Square Planar |
|
sp³d³ |
s + px + py + pz + dxy + dyz + dzx |
Pentagonal Bipyramidal |
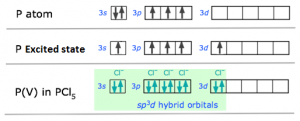
1. sp3d hybridization:
- One s-orbital , three p-orbitals and one d-orbital hybridise to give five degenerate sp3d hybrid orbitals which are then used to form bonds.
- the 5 orbitals are directed towards the corners of a trigonal bipyramidal.
- Orbitals taking part in hybridization: ns, np and nd.
Ex: PCl5
P → 1S², 2S² 2P⁶, 3S² 3Px¹ 3Py¹ 3Pz¹ [Ground State]
P → 1S², 2S² 2P⁶, 3S² 3Px¹ 3Py¹ 3Pz¹ 3d¹ [Ground State]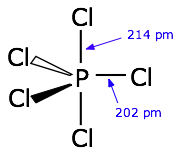
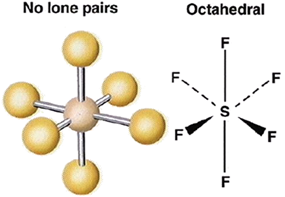
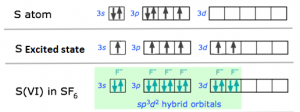
2. sp3d2 hybridization:
- One s, three p and two d orbitals are involved in hybridisation to form the 6 degenerate sp3d2 hybrids.
- The shape of the molecule would be octahedral.
Ex: SF6