S-block:
- The s-block elements are metals.
- Metals are usually solids at room temperature.
- Metals usually have high melting and boiling points. They are good conductors of heat and electricity.
- They are malleable (can be flattened into thin sheets by hammering) and ductile.
P-block:
- The p-block contains metals, metalloids (or semi-metals) and non-metals.
- Non-metals are usually solids or gases at room temperature with low melting and boiling points (boron and carbon are exceptions).
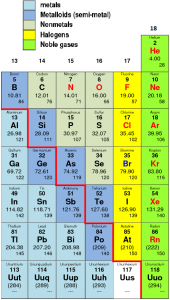
- They are poor conductors of heat and electricity.
- Most nonmetallic solids are brittle and are neither malleable nor ductile.
- The change from metallic to non-metallic character is not abrupt as shown by the thick zig-zag line in.
- The elements (e.g., silicon, germanium, arsenic, antimony and tellurium) bordering this line and running diagonally across the Periodic Table are semi-metals. Their properties are in between those of metals and non-metals.
d-block and f-block:
- The d-block elements are metallic in nature and they form alloys.
- They are said to form covalent bonding as well as the metallic boding.
- The f-block elements, Lanthanides and actinides, are silvery white metals having tensile strength.
