Specific Heat Capacity
Let’s understand the concept of specific heat capacity with the help of examples. Let’s take an equal quantity, say 1 litre, of three liquids. Water, Mustard Oil and Mercury. The concept of specific heat is related to a body’s thermal properties. Let’s heat up these three liquids using the same stove, maintaining the same conditions in all three experiments. The liquids are at a room temperature of 20oC and need to be brought to 60oC. Will all three samples take the same amount of time?
The chances are close to none. While doing the experiment, you will notice that to undergo the same temperature rise, under the same conditions, all three liquids take different times. The mercury is heated up the fastest, followed by Mustard Oil followed then by Water. This property is measured by the heat capacity. The Heat capacity represents the change in temperature in the sample for a given amount of heat. The specific heat in SI Units are joule per kelvin (J kg-1). Specific Heat Capacity which is different from Heat Capacity represents the amount of heat needed to raise the temperature of a unit mass of a substance by 1oC.
What is Specific heat Capacity?
“Specific heat capacity is defined as the energy transferred as heat that is required to raise the temperature of 1 gram of a substance by one kelvin.”
Scientists needed a quantity that has no dependence on the quantity or size of matter under consideration for thermodynamic studies this made them define specific heat capacity. It is an intensive property as it is independent of quantity or size of the matter. Specific heat capacity for any substance or matter can be defined as the amount of heat energy required to raise the temperature of unit mass of that substance by one degree Celsius. Mathematically it is given as:
Q = msΔT
Here Q is the amount of heat energy required to change the temperature of m (kg) of a substance by ΔT, s is the specific heat capacity of the system.
Thermodynamics continues to play a vital role in our lives directly or indirectly. Scientists and engineers use the laws of thermodynamics to design new processes for reactions that would have high efficiency and product yield. Chemical and mechanical engineers apply the concepts of thermodynamics for designing heat engines with high efficiency and better outputs.
Specific Heat Capacity of Water: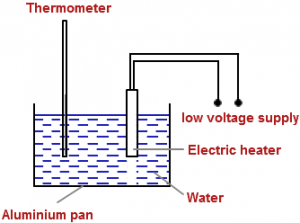 The above diagram shows an experiment for measuring the specific heat capacity of water. The pan contains a measured mass of water. The electric heater is switched on for several minutes and the rise in temperature of the water noted.
The above diagram shows an experiment for measuring the specific heat capacity of water. The pan contains a measured mass of water. The electric heater is switched on for several minutes and the rise in temperature of the water noted.
Assuming that, Thermal energy supplied by heater = thermal energy gained by water
Pt = mc (θ₂ – θ₁)
\(\Rightarrow \,\,C\,\,\,=\,\,\,\frac{Pt}{m\,\,({{\theta }_{2}}\,-\,{{\theta }_{1}})}\).
The specific heat capacity of the water (C) is calculated from this equation by substituting the values of m, t, (θ₂ – θ₁) and P.
How to find the Specific heat Capacity?
Problem: What is the specific heat capacity of iron if it takes 125 J of heat to raise 111 grams by 2.5 degrees Celsius?
Solution: Given,
Q = 125 J
m = 111 grams
ΔT = 2.5⁰ C
Now, we know that:
Q = msΔT
\(\Rightarrow \,\,s\,\,=\,\,\frac{Q}{m\,\,\Delta T}\),
\(\Rightarrow \,\,s\,\,\,=\,\,\,\frac{125\,J}{111\,grams\,\times \,2.5{{\,}^{0}}C}\,\,\,=\,\,\,0.450\,J/g.{{\,}^{0}}C\).
