The Kelvin temperature scale is also known as thermodynamic scale. The S.I. unit of temperature is kelvin and is defined as (1/273.16) of the temperature of the triple point of water. The triple point of water is that point on a P-T diagram where the three phases of water, the solid, the liquid and the gas, can coexist in equilibrium.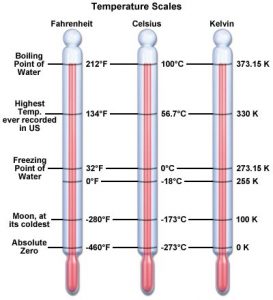 In addition to kelvin temperature scale, there are other temperature scales also like Celsius, Fahrenheit, Reaumer, Rankine etc.
In addition to kelvin temperature scale, there are other temperature scales also like Celsius, Fahrenheit, Reaumer, Rankine etc.
To construct a scale of temperature, two fixed points are taken. First fixed point is the freezing point of water, it is called lower fixed point. The second fixed point is the boiling point of water, it is called upper fixed point.
Temperature on one scale can be converted into other scale by using the following identity.
[Reading on any scale – Lower fixed point (LFP)]/ [Upper fixed point (UFP) – Lower fixed point (LFP)] = Constant for all scales.
C – 0/ 100 = F – 32/ 212 – 32 = K – 273.15/ 373.15 – 273.15.
Or C/5 = F – 32/ 9 =K – 273/ 5.
Where “C” is temperature in Celsius scale, “F” is in Fahrenheit scale and “K” is in Kelvin scale.
