Rate is defined as the change in the amount of the substance or concentration of the substance with respect to time.
\(RATE\text{ }=\text{ }\frac{\left[ Concentration \right]}{Time}\).
Let us consider a general reaction,
Reactants → Products
During the progress of a chemical reaction,
Concentration of the reactants are decreases with time and concentration of products increases with time.
The following image shows diagrammatic representation of RATE OF REACTION.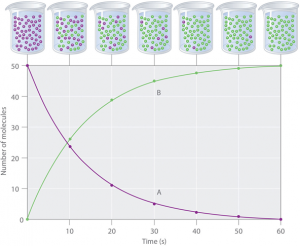 In above chemical reaction,
In above chemical reaction,
- Represents the REACTANT molecules
- Represnts the PRODUCT molecules
Rate of a reaction for chemical reactions,
\(RATE=-\frac{[\operatorname{Re}ac\tan ts]}{Time}=+\frac{[products]}{Time}\).
UNITS OF RATE OF REACTION:
Units = Concentration.time¯¹
= Mole.Lit¯¹.time¯¹
= Mole.dm¯³.time¯¹
Time can be expressed in terms of Years, Months, Days, Hours, Minutes, and Seconds.
