These are governed by the Faraday’s laws of electrolysis
First law of Electrolysis:
The amount of chemical change produced is proportional to the quantity of electric charge passing through an electrolytic cell. Greater the number of moles of electrons transferred, the greater the mass of the products.
W α Q (charge)
Charge = (current) ✕ time
W = Zit
W = Mass of the metal deposited
= Gas liberated due to passage of current.
i = current
t = time
Anode:
½ H₂O → ½ O₂(g) + H²⁺(g) + e¯
Cathode:
½ Cu²⁺(aq) + e¯ → ½ Cu(s)
From these half – cell reactions we would define one (electrochemical) equivalent as equal to 0.5 mole H₂O, 1 mole of H⁺, 0.5 mole of Cu²⁺, and 0.5 mole of Cu(s)
Second law of electrolysis:
A given Quantity of electricity produces the same number of equivalents of any substance on electrolysis.
Mass of metal A/ Mass of metal B = Equivalent weight of A/Equivalent weight of B
The chemical equivalent of an element is numerically equal to its relative atomic mass in grams divided by the valency of the ion.
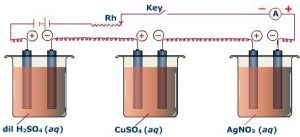
If we take three solutions of electrolytes: AgNO3, CuSO4 and Al(NO3)3 in a series, and pass some quantity of electricity through them for the same time then, Ag, Cu and Al metals collect at the cathode. Their masses are directly proportional to their equivalent masses.
Equivalent Mass = Atomic mass of element/ Valency of element
According to Faraday, if 96,500 Coulombs (or 1 Faraday) is passed through these electrolytes,
we get Ag = 108/1 = 108g; Cu = 63.5/2 = 31.75g; Al = 27/3 = 9g which are the equivalent masses of Ag, Cu and Al respectively.
