⇒ Phosphorus forms a large number of oxoacids.
⇒ All these acids are based on tetrahedral four coordinated phosphorus atom containing at least one P = O unit and one P – OH group. Condensed systems are formed by P – O – P linkage or P – P linkage.
⇒ The basicity depends on the H atoms bonded to O in the form of – OH.
Some of the common oxoacids of phosphorus are as follows:
|
Name |
Formula | Oxidation state of P | basicity | structure |
preparation |
|
Hypophosphorus acid |
H3PO2 | +1 | 1 | 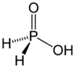 |
white P4 + alkali |
|
Phosphorus acid |
H3PO3 | +3 | 2 | 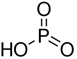 |
P2O3 + 3H2O → 2H3PO3 |
|
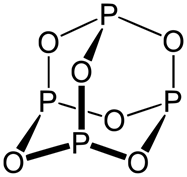
Hypophosphoric acid |
H4P2O6 | +4 | 4 | 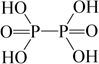 |
red P4 + alkali |
|
Orthophosphoric acid |
H3PO4 | +5 | 3 | 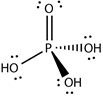 |
P(red) + 5HNO3 → H2O + H3PO4 + 5NO2 |
|
Diphosphoric acid (Pyrophosphoric acid) |
H4P2O7 | +5 | 4 | 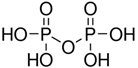 |
heating phosphoric acid 2H3PO4 → H4P2O7 + H2O |
|
Metaphosphoric acid |
HPO3 | +5 | 1 | 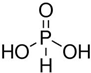 |
heating orthophosphoric acid to about 850 K H3PO4 → HPO3 + H2O |
|
Peroxophosphoric acid |
H3PO5 | +7 | 3 | 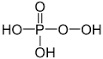 |
P2O5 + 2H2O2 + H2O → 2H3PO5 |
Oxides of phosphorous:
1. Phosphorus pentoxide:
⇒ It is a chemical compound with molecular formula P4O10 (with its common name derived from its empirical formula, P2O5).
⇒ This white crystalline solid is the anhydride of phosphoric acid. It is a powerful desiccant and dehydrating agent.
⇒ P4O10 is prepared by burning elemental phosphorus with sufficient supply of air:
P4 + 5 O2 → P4O10
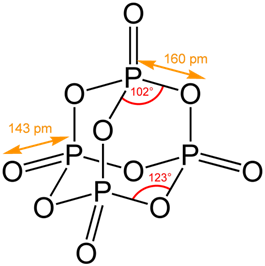
2. Phosphorus trioxide:
⇒ It is the chemical compound with the molecular formula P4O6. Although it should properly be named tetraphosphorus hexoxide, the name phosphorus trioxide preceded the knowledge of the compound’s molecular structure, and its usage continues today.
⇒ It is a colorless solid. It is formally the anhydride of phosphorous acid, H3PO3, but cannot be obtained by the dehydration of the acid. It is a white, waxy, crystalline and highly toxic solid.
⇒ It is obtained by the combustion of phosphorus in a limited supply of air at low temperature.
P4(s) + 3 O2 (g) → P4O6(s)