Defects in Crystals: In a perfect crystal all the particles are present at their respective positions. However, no crystal is perfect, every crystal is associated with some deviations. These deviations are called defects (or) imperfections.
These are mainly two types.
- Stoichiometric defects
- Non- Stoichiometric defects
Stoichiometric defects: The compounds in which ratio between number of cations and number of anions is exactly to the ratio indicated by the formula are called Stoichiometric compounds.
These are the following types:
- Schottky defect
- Frenkel defect
- Interstitial defects
- Vacancy defects
1. Schottky defect:
⇒ This is arises due to missing of set of cations and anions leaving behind a pair of Holes.
⇒ This is observed in ionic compounds with high coordination numbers.
⇒ This is observed in ionic compounds in which the sizes of anions and cations are almost equal.
Ex: Nacl, KCl, NaBr, CsCl etc….
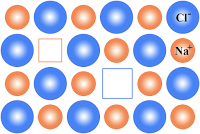
Consequences of Schottky defect:
⇒ As the number of ions decreases as a result of this defect, the mass decreases whereas the volume remains the same. Hence density of the solid decreases.
⇒ The crystal begins to conduct electricity to a small extent by ionic mechanism.
⇒ The presence of too many voids lowers lattice energy and the stability of the crystal.
2. Frenkel Defect: This arises due to missing of an ion from its correct lattice site and occupies an interstitical site.
This is observed mainly in:
⇒ Ionic compounds with low coordination number.
⇒ Ionic compounds in which sizes of cations and anions differ largely.
Ex: AgCl, AgBr, AgI, ZnS etc……..
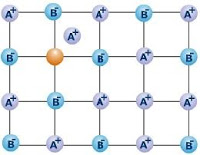
Consequences of Frenkel defect:
⇒ As no ions are missing from the crystal lattice as a whole, therefore density of the solid remains the same.
⇒ The closeness of like charges tends to increases the dielectric constant of the crystal.
⇒ The crystal conducts electricity to a small extent by ionic mechanism.
3. Interstitial defect:
⇒ This type of defect is caused due to the presence of ions in the normally vacant interstitial sites in the crystals.
⇒ Interstitial impurity atoms are much smaller than the atoms in the bulk matrix.
⇒ Interstitial impurity atoms fit into the open space between the bulk atoms of the lattice structure.
⇒ An example of interstitial impurity atoms is the carbon atoms that are added to iron to make steel. Carbon atoms, with a radius of 0.071 nm, fit nicely in the open spaces between the larger (0.124 nm) iron atoms.
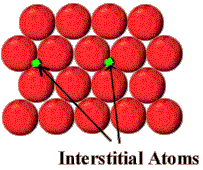
4. Vacancy defect:
⇒ Vacancies are empty spaces where an atom should be, but is missing.
⇒ They are common, especially at high temperatures when atoms are frequently and randomly change their positions leaving behind empty lattice sites.
⇒ In most cases diffusion (mass transport by atomic motion) can only occur because of vacancies.

