Electric Charge
Electric charge is the property of sub-atomic particles particularly includes electrons and protons. Electrons have negative charge, protons have positive charge and neutrons do not have any charge. Or it is a physical property of matter which causes the force when places in any electromagnetic field.
What is Electric Charge?
Electric Charge is defined as the amount of energy or electrons passing from one body to another either by conduction, induction or other specific methods. There are two types of electric charges namely positive charge and negative charges. Charges are present in almost everybody and those bodies which don’t have charges are assumed to be neutrally charged or no charge.
The charge is denoted by the symbol ‘q’ and its standard unit is coulomb.
q = ne
Where,
q = Charge,
n = Number of Electrons,
e = Charge on one electron = 1.6 x 10⁻¹⁹ C.
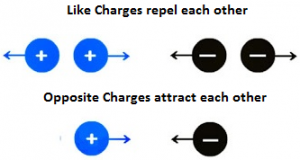
Basic Nature of Charges: The two very basic nature of electric charges is,
- Like charges repel each other that is one electron will repel another electron, or one proton will repel another proton.
- Unlike charges attract each other, that one electron will attract another proton or vice versa.
The nature of charge is responsible for the force acting on them and coordinating direction of flow of them. The charge on electron and proton is same in magnitude which is 1.6 × 10-19 C. The only difference is that we show charge on proton with a positive sign and on electron with a negative sign.
