- When mono substituted benzene is subjected to further substitution, di-substituted products are formed. They are Ortho, para and meta.
- The product which is formed more predominately depends on nature of the substituents already present in the benzene ring.
- This is known as direct influence of substituents.
Ortho and para directing groups:
- They direct the incoming group to ortho and para positions. It is due to the increase in electron density at ortho and para position.
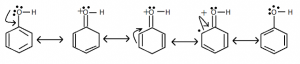 Ring activating groups are – NH₂, – NHR, – NHCOCH₃, – OCH₃ – CH₃, C₂H₅ etc…
Ring activating groups are – NH₂, – NHR, – NHCOCH₃, – OCH₃ – CH₃, C₂H₅ etc… - Halogens are ring deactivating group because of – I effect. But due to resonance the electron density at 0 and P – position is greater than at meta-position. Hence it is O – and p – directing group.
Meta-directing group:
- These groups direct the coming group to meta-position.Ex: – NO₂, – CN, – CHO, -COR, – COOH, – COOR, – SO₃H etc…
- These groups reduces the electron density in the benzene ring especially at O – and p – position.
- Therefore further substitution stakes place at meta-position.
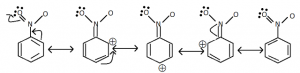
- Therefore these groups are called deactivating groups.
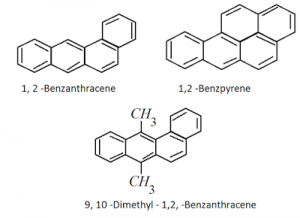
Carcinogenicity and Toxicity:
- Benzene and polynuclear hydrocarbons containing more than two benzene rings fused together are toxic and they possess cancer producing (carcinogenic) property. These are formed by incomplete combustion of organic materials like Tobacco, coal and petroleum.
- These undergo various biochemical reactions that damage DNA and causes cancer.
Ex: