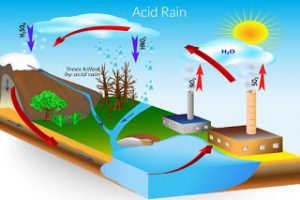
• Acid rains occur due to presence of oxides of nitrogen & Sulphur in rain water.
• Nitric acid & sulphuric acid in the atmosphere dissolve in rain water and come down to earth as acid rain.
• The rain water which contains nitric acid and sulphuric acid is called acid rain.
• Acid rain is more in industrial area.
• Acid rain is reported in some places where there are no industries.
• This is due to movement of rain clouds from industrial area to other areas due to the flow of wind.
• Acid rain was reported at Delhi, Calcutta, Surat, Mumbai and Hyderabad in India.
• Acid rain may have a pH of 4 – 5.
• Acid rains corrode monuments, buildings, statues, bridges & marble stones.
• The glossy nature of Tajmahal is affected due to acid rain.
• They destroy forests and crops affecting the fertility of soil and reduce the agriculture productivity.
H2O (l) + CO2 (g) → H2CO3 (aq)
NO + O3→ NO2 + O2
NO2 + O3 → NO3 + O2
NO2 + NO3 → N2O5
N2O5 + H2O → 2HNO3
\(S{{O}_{2}}+\frac{1}{2}{{O}_{2}}\to S{{O}_{3}}\xrightarrow{{{H}_{2}}O}{{H}_{2}}S{{O}_{4}}\)\(S{{O}_{2}}+\frac{1}{2}{{O}_{2}}+{{H}_{2}}O\xrightarrow[Metal\text{ }or\text{ }soot\text{ }particles]{NO}{{H}_{2}}S{{O}_{4}}\)
Particulates:
Particulates in the atmosphere may be viable or non¬viable.
• Viable are minute living organisms that are dispersed in the atmosphere. Example: bacteria, fungi, moulds, algae etc.
• Non¬viable particulates may be classified as:
a. Smoke particulates: consist of solid or mixture of solid and liquid particles formed during combustion of organic matter. Example: cigarette smoke, smoke from burning of fossil fuel, garbage and dry leaves, oil smoke etc.
b. Dust: composed of fine solid particles (over 1μm in diameter), produced during crushing, grinding and attribution of solid materials.
Sand from sand blasting, saw dust from wood works, pulverized coal, cement and fly ash from factories, dust storms etc., are some typical examples of this type of particulate emission.
c. Mists: Are produced by particles of spray liquids and by condensation of vapours in air. Example: sulphuric acid mist and herbicides and insecticides that miss their targets and travel through air and form mists.
d. Fumes: Are generally obtained by the condensation of vapours during sublimation, distillation, boiling and several other chemical reactions. Generally, organic solvents, metals and metallic oxides form fume particles.
Smog:
• Smog is a mixture of smoke and fog.
• Smog formed during summer is oxidizing (photochemical smog) and that formed during winter is reducing type and contains smoke, fog and Sulphur dioxide.
• The exhaust fumes released by motor vehicles include a mixture of hydrocarbons like
methane, ethane, acetylene, propane, butane.
Photo chemical smog:
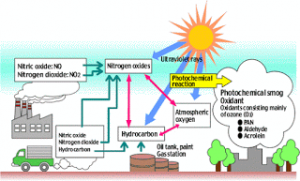
• Hydrocarbons present in the atmosphere undergo photochemical oxidation and mix with oxides of nitrogen to form photochemical smog.
• O3 and NO2 being strong oxidising agents react with hydrocarbons producing chemicals formaldehyde, acrolein and peroxy acetyl nitrate (PAN) which are harmful ingredients of photochemical smog.
NO2 (g) + energy from sun light → NO (g) + O (g)
O2 (g) + O (g) → O3 (g)
NO (g) + O3 (g) → NO2 (g) + O2 (g)
3CH4 + 2O3 → 3HCHO + 3H2O
\(C{{H}_{2}}=CH-CHO\xrightarrow{{{O}_{3}},N{{O}_{2}}}C{{H}_{3}}CO-OON{{O}_{2}}\)(PAN)
• The main constituents of photochemical smog are peroxides, aldehydes, ketones and organ nitrates.
Effects:
• Irritates eyes & lungs.
• Causes asthma & bronchitis.
• Causes cracking of rubber and extensive damage of plant life.
• Reduces the visibility & causes corrosion of metals, stones & building material, paper, leather & painted surface.
