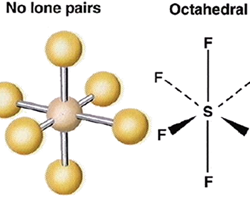
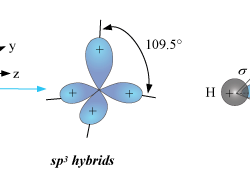
Applications of Molecular Orbit Theory
Electronic configuration and Molecular Behavior: The distribution of electrons among various molecular orbitals is called the electronic configuration of the molecule. Important information of configuration: a) Stability of Molecules: The molecule is stable if NB is greater than NA The molecule is unstable if NB is less than NA. Where NB → Bonding e⁻s NA Read more about Applications of Molecular Orbit Theory[…]