Hello Guys…!!
Here is a fast over-view of Laws of Thermodynamics.
Zeroth Law of Thermo dynamics:
Let A, B, C be different bodies having different temperatures. If heat exchange is possible between A and B and B and C then it is also possible between A and C.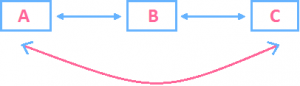 Work and Heat:
Work and Heat:
Work:
- It is the mode of energy transfer mechanical work = PΔV = Pext (V₂ – V₁).
- It is a path dependent function
- Work done on system increases the energy of a system and work done by system decreases the energy of system
Heat:
- It is the quantity of heat which flows between system and surrounding.
- Always flows from higher temperature to lower temperature.
Internal Energy:
E = Etranslational + Erotational + Evibrational + Ebonding + …
Summation of all types of energies constitutes to give net internal energy of a particular system.
First law of thermo dynamics:
Energy can neither be created nor be destroyed. It can be only transformed from one form to another.
Total energy of the universe is constant.
Whenever a quantity of one king of energy disappears, an exactly equivalent quantity of energy in some other form must appear.
E₂ = E₁ + q + W
[E₂ – E₁ = q + W]
ΔE = q + W → (First Law)
Enthalpy:
Heat content at constant pressure is called “Enthalpy”.
ΔH = ΔE + PΔV
Heat capacity:
Quantity of heat required to raise the temperature of the system by one degree.
Heat capacity = \(\left( \frac{dq}{dT} \right) \)
Heat capacity at constant volume = \({{C}_{v}}={{\left( \frac{dE}{dT} \right)}_{v}}\)
Heat capacity at constant pressure = \({{C}_{p}}={{\left( \frac{dH}{dT} \right)}_{P}}\)
Cp – Cv = R
Expansion of an Ideal gas:
1. Isothermal expansion:
ΔE = 0 → for isothermal Process
ΔE = q + w → from first law
∴ q = – ω
ΔH = ΔE + PΔV
ΔH = ΔE + nRΔT
∴ ΔH = 0
a) Work done in reversible isothermal process:
Pext = Pgas = P
\(W=\int\limits_{-{{v}_{1}}}^{{{v}_{2}}}{Pdv}\)\(W=\int\limits_{-{{v}_{1}}}^{{{v}_{2}}}{\frac{nRT}{V}dv}\)
\(W=-nRT\,\ln \left( \frac{{{V}_{2}}}{{{V}_{1}}} \right) \)b) Work done for irreversible process: W = – Pext (V₂ – V₁)
2. Adiabatic process:
q = 0
ΔE = q + ω
∴ ΔE = ω for an adiabatic process.
Important results for adiabatic process:
- PVϒ = Constant
- TVϒ⁻¹ = Constant
- PϒT¹⁻ϒ = Constant
Work done for reversible adiabatic process
\(W=\frac{nR}{(\Upsilon -1)}({{T}_{2}}-{{T}_{1}})\)
Work done for irreversible adiabatic process
\(W=-R{{P}_{ext}}\left( \frac{{{T}_{2}}{{P}_{1}}-{{T}_{1}}{{P}_{2}}}{{{P}_{1}}{{P}_{2}}} \right) \)
Second Law of Thermodynamics:
The Second Law of Thermodynamics states that the state of entropy of the entire universe, as a closed isolated system, will always increase over time. The second law also states that the changes in the entropy in the universe can never be negative.
- Second law of thermodynamics gives rise to the concept of Entropy
- Entropy (S) -measure of the number of specific ways in which a thermodynamic system may be arranged, commonly understood as a measure of disorder.
\(n{{C}_{p}}\,\ln \left( \frac{{{T}_{2}}}{{{T}_{1}}} \right)+nR\,\ln \left( \frac{{{P}_{1}}}{{{P}_{2}}} \right) \)
Gibb’s Free Energy (G):
G = H – TS
ΔG = ΔH – TΔS at constant temperature.
Also, dG = Vdp – SdT
At constant temperature, \(\Delta G=nRT\,\ln \left( \frac{{{p}_{2}}}{{{p}_{1}}} \right) \)
Criteria for Spontaneity:
For a spontaneous process, entropy of universe must increase, i.e. ΔSuniv > 0 or ΔG < 0
(i) If ΔH < 0 and ΔS > 0, ΔG is always negative, always spontaneous.
(ii) If ΔH > 0 and ΔS < 0, ΔG is always positive, always non-spontaneous
(iii) If ΔH > 0 and ΔS > 0, process will be spontaneous higher temperature but non-spontaneous at lower temperature.
(iv) If ΔH < 0 and ΔS < 0, process will be spontaneous lower temperature but non-spontaneous at higher temperature.
