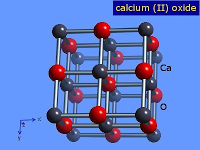
1. Calcium oxide (Quick Lime)
- Lime stone found in nature is the starting material of almost all the compounds of calcium.
- Calcium oxide is also called Quick line.
- The decomposition of calcium carbonate is a reversible process
CaCO₃ ⇋ CaO + CO₂
- When water is added to lime, a hissing sound is produced along with clouds of steam. In this process a large amount of heat energy is given out.
CaO + H₂O → Ca (OH)₂ + Heat
Quick lime Slaked Lime Energy
Uses of Quick lime:
- As it has high melting point (260⁰C) it is used for furnace lining.
- It is used as a basic flux in metallurgy as it combines with acidic impurities.
CaO + SiO₂ → CaSiO₃ - It is used as disinfectant and germicide, as a drying agent for ammonia and alcohol.
- It is employed for the purification of coal gas and softening of water.
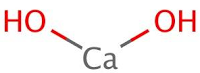
2. Lime water: [Ca (OH)₂] [Calcium Hydroxide]
- The chemical formula is Ca(OH)₂
- When water is added to lime, a hissing sound is produced along with clouds of steam in this process a large amount of heat energy is given out. The lime cracks with the formation of a powder called slacked lime Ca (OH)₂.
CaO + H₂O → Ca(OH)₂ + Heat
Quick lime Slaked Lime Energy - The above process is known as slaking of lime
Uses of slacked lime: [Ca (OH)₂]
- Slaked lime is applied to acidic soils to regulate the soil’s pH levels.
- This allows for faster growth of crops by aiding the intake of nutrients and nitrogen by vegetation growing in the soil.
- Calcium hydroxide is also used as a filler material in rubber and plastics, serving as an acceleration in the rubber.
3. Calcium carbonate: [CaCO₃]
- It is found in nature in huge quantities in various forms such as lime stone, marble, chalk.
- It can be obtained by passing carbon dioxide through lime water (or) by adding sodium carbonate solution to CaCl₂.
Ca (OH)₂ + CO₂ → CaCO₃ + H₂O
CaCl₂ + Na₂CO₃ → CaCO₃ ↓ + 2NaCl - The precipitate of CaCO₃ thus obtained is known as precipitated chalk.

- It is a white powder, insoluble in water. It dissolves in presence of CO₂ due to formation of calcium bicarbonate.
CaCO₂ + H₂O + CO₂ →Ca (HCO₃)₂
Uses of CaCO₃
- Precipitated chalk is used in tooth pastes and face powders, in medicine for indigestion, in adhesives and in cosmetics.
- Chalk is used in paints and distempers.
- Marble is used in for building purposes, production of CO₂ in laboratory.
- Lime stone is used:
i) In the manufacture of the quick lime, slacked lime, cement, washing soda and glass.
ii) Used as flux in the smelting of iron and lead ores.
4. Plaster of Pairs: [2CaSO₄ H₂O] [Calcium sulphate dihydrate]
Preparation:
It is obtained when gypsum, calcium sulphate dihydrate [2CaSO₄ 2H₂O] is heated at 120⁰C
2[CaSO₄ 2H₂O] → 2CaSO₄ H₂O + 3H₂O
Plaster of Paris
Properties:
- Plaster of Paris is a white powder
- It has the property of setting to a hard mass when a paste with water is allowed to stand aside for some time. Slight expansion occurs during the setting as water is absorbed to reform CaSO₄ 2H₂O (gypsum)
The setting takes place in two stages:
In the first stage, there is conversion of Paris into orthorhombic form of gypsum and in the second stage orthorhombic form changes into monoclinic form.
\(2CaS{{O}_{4}}.{{H}_{2}}O\xrightarrow[{{H}_{2}}O]{setting}CaS{{O}_{4}}.2{{H}_{2}}O\xrightarrow{Hardenning}CaS{{O}_{4}}.2{{H}_{2}}O\,\,\left( Mono\,\,clinic \right)\)Uses:
- It is used in surgery for setting broken (or) dislocated bones.
- In making casts for statue, toys, surgical instruments etc…
- In making casts in dentistry.
- In making black board chalk
5. Portland cement:
- Cement is one of the most important building materials at the present time. It is used in the construction of buildings, roads, bridges, dams.
- Cement is a dirty greenish heavy powder containing calcium aluminates and silicates.
Composition of Portland cement:
|
Calcium Oxide |
(CaO) |
61.5% |
|
Silica |
(SiO₂) |
22.5% |
|
Alumina |
(Al₂O₃) |
7.5% |
|
Magnesium Oxide |
(MgO) |
2.5% |
|
Ferric Oxide |
(Fe₂O₃) |
2.0% |
|
Potassium Oxide |
(K₂O₃) |
1.5% |
|
Sulphur Oxide |
(SO₃) |
1.0% |
Raw Material:
The important Raw materials needed for the manufactures of cement are
- Lime stone – The provides lime
- Clay – This provides alumina and silica
- Gypsum
Manufacture of Cement:
- Dry Process
- Wet Process
Dry Process:
- The process is used when lime stone is hard in nature.
- The lime stone is first broken into small pieces.
- It is then mixed with clay in proper proportions.
- The mixture is finally pulverized to such an extent that it possess through a 100 mesh sieve.
- This homogeneous mixture is known as raw material.
Wet Process:
- The process is used when lime stone and clay both are soft in nature.
- The clay is washed with water in wash mill to remove foreign materials like flint.
- The powdered lime stone is mixed with clay paste in the ratio of 75% (Lime Stone) and 25% (Clay).
- The mixture is finely ground and made homogeneous paste is known as surely. It contains 40% of water.
