CARBENES
Carbenes are highly reactive molecule containing a divalent carbon atom.
Neutral, divalent, highly reactive in nature. They are highly unstable that’s why highly reactive in nature. Simplest carbene is \(\overset{\bullet \bullet }{\mathop{C{{H}_{2}}}}\,\)
General preparation: Carbenes are related to carbanions through α – elimination reaction
\(CH{{X}_{3}}\xrightarrow{alcol\,KOH/\Delta }\underset{Di\,halo\,Carbene}{\mathop{\underset{\downarrow }{\mathop{\overset{\bullet \bullet }{\mathop{C{{X}_{3}}}}\,+H\oplus }}\,}}\,\)
Hybridization of the divalent carbon may be sp² as well as sp. sp² hybrid carbine is of 2 types
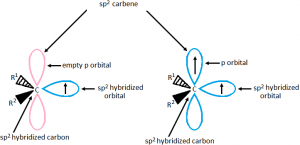
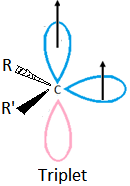
sp – hybrid carbine is always triplet:
The decreasing order of stability of different types of singlet carbenes is as follows:
\(\overset{\bullet \bullet }{\mathop{C}}\,{{H}_{2}}>\overset{\bullet \bullet }{\mathop{C}}\,C{{l}_{2}}>\overset{\bullet \bullet }{\mathop{C}}\,{{F}_{2}}\)
Carbenes can exist in singlet as well as in triplet state but \(\overset{\bullet \bullet }{\mathop{C}}\,{{X}_{2}}\) exists only in single state.
Important points regarding carbene Reaction: Mainly give two types of reactions.
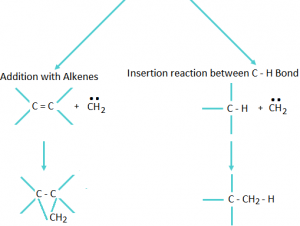
Important points about Carbene: Unusual carbene containing species.
It has a carbon with a lone pair and an empty orbital. The empty orbital makes the carbon very reactive \(\overset{\bullet \bullet }{\mathop{C{{H}_{2}}}}\,\) is the simplest carbene (methylene). Obtained by heating diazo methane.
\(\,\,\overset{-}{\mathop{C{{H}_{2}}}}\,-\overset{\oplus }{\mathop{N}}\,\equiv N\xrightarrow[C{{H}_{2}}-\,C{{H}_{2}}]{\Delta }\underset{Cyclopropane}{\mathop{\Delta }}\,+\,\overset{\,\!:}{\mathop{C{{H}_{2}}}}\,\)Nitrene:
As carbene is for carbon nitrene is for nitrogen. It is analogues of carbene. There is possibility of two spin states for a nitrene depending on whether the two non-bonding electrons have their spins paired or parallel. They are highly reactive because they are highly unstable and always form as a reaction intermediate.
Nature:
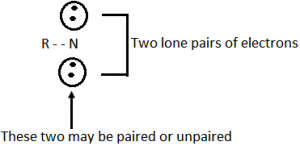
In general, nitrene obeys Hund’s rule and ground state triplet with two degenerated sp – orbitals containing a single electron each.
