Adsorption isotherms: Plot of the amount of gas adsorbed on the surface of the adsorbent and pressure at constant temperature.
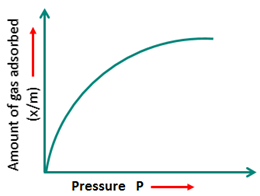 1. Freundlich adsorption isotherm:
1. Freundlich adsorption isotherm:
\(\frac{x}{m}=k.{{P}^{\frac{1}{n}}}\) (n > 1) … 1
x = mass of the gas adsorbed
m = mass of the adsorbent
P = pressure
k, n = constants. Depend on the nature of the adsorbent and the gas
Taking logarithm of eqn 1 →\(\log \left( \frac{x}{m} \right)=\log k+\frac{1}{n}\log P\)
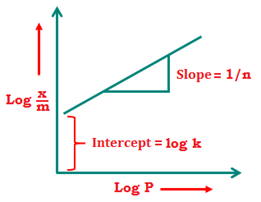
The plot of log(x/m) and log P is a straight line. If the plot is not a straight line then Freundlich isotherm is not valid. The slope of the straight line is 1/n and the y intercept is equal to k.
If 1/n = 0, then x/m is constant and doesn’t depend on pressure
If 1/n = 1, then x/m α P
2. Langmuir adsorption isotherm:
Postulates:
⇒ Gases undergoing adsorption behave ideally
⇒ The surface containing the adsorbing sites is perfectly flat plane with no corrugations (assume the surface is homogeneous).
⇒ The adsorbing gas adsorbs into an immobile state.
⇒ All sites are equivalent.
⇒ Each site can hold at most one molecule of gas (mono-layer coverage only).
⇒ There are no interactions between adsorbate molecules on adjacent sites.
⇒ Dynamic equilibrium exists between adsorbed gaseous molecules and the free gaseous molecules
![]()
Where A(g) is unabsorbed gaseous molecule, B(s) is unoccupied metal surface and AB is Adsorbed gaseous molecule. He gave the following relation:
x\(\frac{P}{x/m}=\frac{1}{{{K}’}}+\left( \frac{K}{{{K}’}} \right)P\Rightarrow \frac{x}{m}=\frac{{K}’P}{1+KP}\)
\(K=\frac{{{k}_{ad}}}{{{k}_{d}}}\), kad = adsorption rate constant, kd = description rate constant
K’ = kK
The plot of x\(\frac{P}{x/m}\) and P is a straight line, whose slope is (K/K’) and y intercept is (1/K’)
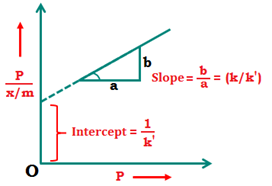
When pressure is very high, then 1 + KP = KP
Thus, at high pressures the degree of adsorption approaches a limiting value.
When pressure is low then 1 + KP = 1
\(\frac{x}{m}={K}’P\)
The degree of adsorption is directly proportional to pressure
Adsorption from solution phase:
⇒ The adsorption decreases with increase in temperature.
⇒ Adsorption increases with increase in surface area.
⇒ The extent of adsorption depends on concentration of solute in solution.
⇒ The extent of adsorption depends on nature of adsorbent and adsorb ate.
The Freundlich equation is modified as \(\frac{x}{m}=k.{{C}^{\frac{1}{n}}}\)
C – Equilibrium concentration
\(\log \left( \frac{x}{m} \right)=\log k+\frac{1}{n}\log C\)
Positive adsorption: The concentration of adsorbate is more on the surface of adsorbent than in the bulk of the solution
Negative adsorption: The concentration of adsorbate is more in the bulk of the solution than in the surface of the adsorbent.
